Saadi D., Mayr F., Yumusak C., Wielend D., Cobet M., Kahraman B., Irimia C.V., Kanbur Y., Bednorz M., Kotwica K., Ben Fredj A., Romdhane S., Scharber M.C., Sariciftci N.S., Irimia-Vladu M. N,N′-Substituted quinacridones for organic electronic device applications
Materials Advances, 2023, Article in Press
DOI: 10.1039/d2ma01010k
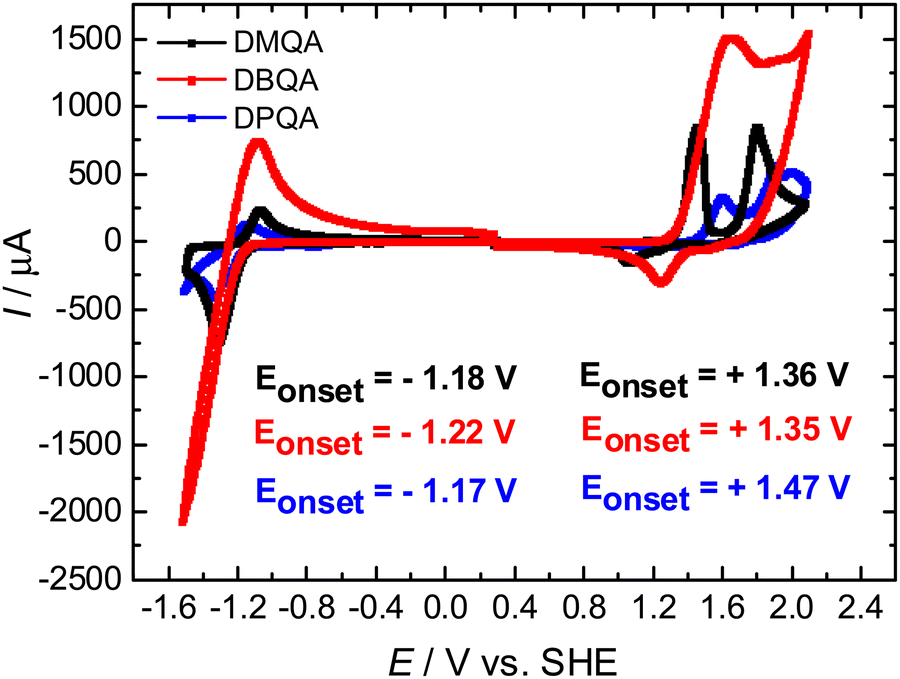
Abstract:N,N′-Substituted quinacridones are a novel class of commercially available quinacridones for organic electronics which are reported here. In this study, we performed in-depth investigations of the material properties of these molecules i.e. their optical and charge transport properties, infrared-active vibrations (FTIR), electrochemical reduction and oxidation properties, thin film forming and processability, and finally performance in organic field effect transistor devices. We show that substitution plays a critical role in the charge transport properties, with methyl substituted amine being the most favorable, followed by di-phenyl and finally di-butyl. © 2023 The Author(s).
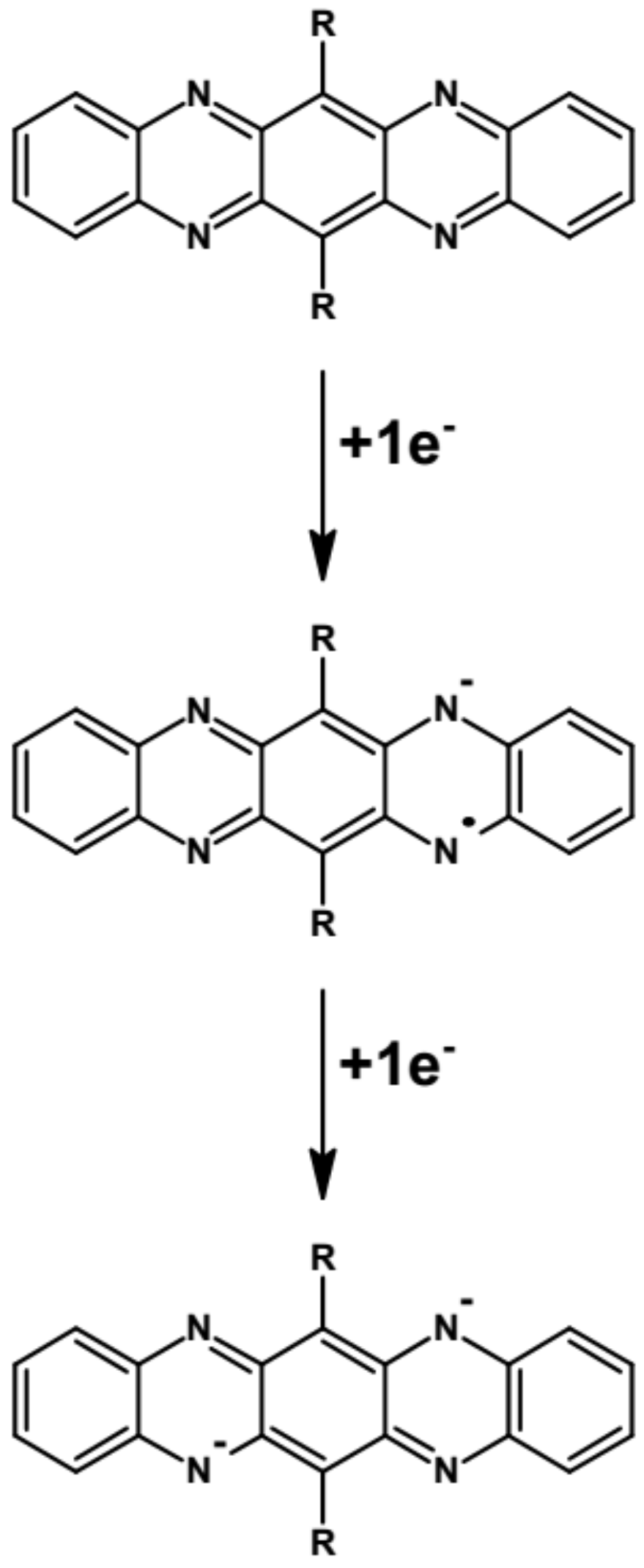
Kulszewicz-Bajer I., Guzauskas M., Makowska-Janusik M., Zagórska M., Mahmoudi M., Grazulevicius J.V., Proń A., Volyniuk D. Acridone and quinacridone derivatives with carbazole or phenoxazine substituents: synthesis, electrochemistry, photophysics and application as TADF electroluminophores
Journal of Materials Chemistry C 2022, 10, 34, 12377-12391
DOI: 10.1039/d2tc02270b
Abstract:Six acridone (quinacridone) derivatives containing either carbazole or phenoxazine substituents were designed and synthesized with the aim of elucidating the effect of the donor (D) and acceptor (A) linking pattern (D-A, D-A-D or D-π-A-π-D) on their photophysical and electrochemical properties. These new electroactive compounds combine reversible electrochemical oxidation with excellent luminescent properties. Their electrochemically determined ionization potentials (IPs) are in the range from 5.09 eV to 5.45 eV, higher for derivatives with carbazole donors as compared to phenoxazine ones. The measured electron affinities (EAs) are in the range from −2.53 eV to −2.64 eV with the exception of the quinacridone derivative showing EA of −3.03 eV. Their vacuum-deposited films emit radiation in a wide spectral range from sky-blue to red. Compounds with carbazole moieties (compounds 1, 2 and 6 in the subsequent text) showed prompt fluorescence and aggregation-caused quenching. Photoluminescent quantum yields (PLQYs) of their toluene solutions reached values up to 69%. Compounds containing phenoxazine moieties (compounds marked as 3-5) demonstrated thermally activated delayed fluorescence (TADF) and aggregation-induced emission enhancement (AIEE). Their neat films showed PLQYs of 35%. Quinacridone disubstituted with carbazole (compound 6) showed the highest hole mobility reaching 2.53 × 10−3 cm2 V−1 s−1 at electric field of 3.6 × 105 V cm−1. Carbazolyl disubstituted acridone (compound 2) and phenoxazinyl monosubstituted acridone (compound 3) turned out to be ambipolar compounds showing reasonably balanced electron and hole mobilities. The appropriate combination of redox, transport and luminescent properties makes the studied compounds suitable candidates for optoelectronic applications. Test OLEDs fabricated from 3 exhibited maximum external quantum efficiencies reaching 16.7%. Finally, an excellent agreement between the experimental results and those obtained by DFT calculations should be stressed. The basics for selection according to the user needs of either D-A, D-A-D or D-π-A-π-D types of molecular structures of TADF/AIEE luminophores are provided in this study. © 2022 The Royal Society of Chemistry.
Rybakiewicz-Sekita R., Toman P., Ganczarczyk R., Drapala J., Ledwon P., Banasiewicz M., Skorka L., Matyjasiak A., Zagorska M., Pron A. D-A-D Compounds Combining Dithienopyrrole Donors and Acceptors of Increasing Electron-Withdrawing Capability: Synthesis, Spectroscopy, Electropolymerization, and Electrochromism
Journal of Physical Chemistry B 2022, 126, 22, 4089-4105
DOI: 10.1021/acs.jpcb.2c01772
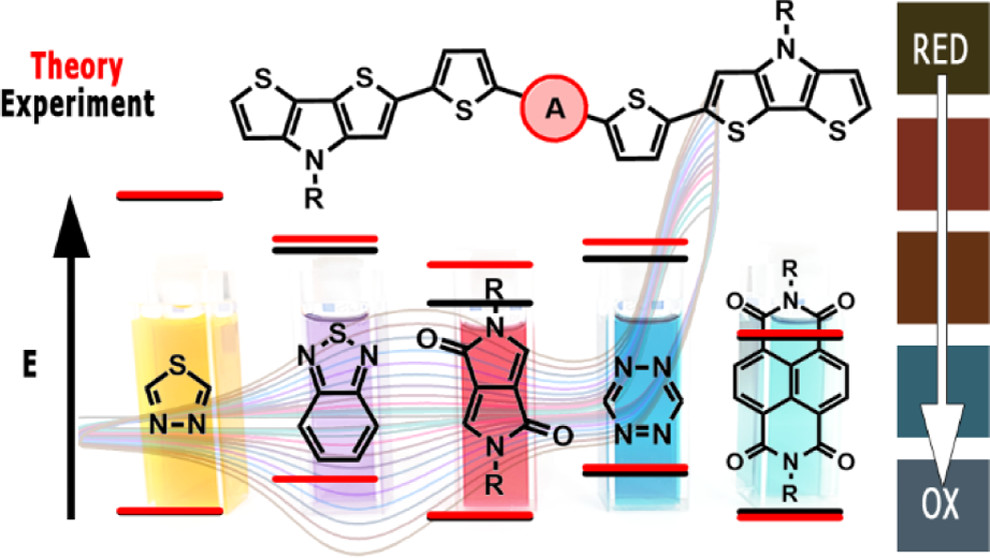
Abstract: Five D-π-A-π-D compounds consisting of the same donor unit (dithieno[3,2-b:2′,3′-d]pyrrole, DTP), the same π-linker (2,5-thienylene), and different acceptors of increasing electron-withdrawing ability (1,3,4-thiadiazole (TD), benzo[c][1,2,5]thiadiazole (BTD), 2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione (DPP), 1,2,4,5-tetrazine (TZ), and benzo[lmn][3,8]phenanthroline-1,3,6,8(2H,7H)-tetraone (NDI)) were synthesized. DTP-TD, DTP-BTD, and DTP-DPP turned out to be interesting luminophores emitting either yellow (DTP-TD) or near-infrared (DTP-BTD and DTP-DPP) radiation in dichloromethane solutions. The emission bands were increasingly bathochromically shifted with increasing solvent polarity. Electrochemically determined electron affinities (|EA|s) were found to be strongly dependent on the nature of the acceptor changing from 2.86 to 3.84 eV for DTP-TD and DTP-NDI, respectively, while the ionization potential (IP) values varied only weakly. Experimental findings were strongly supported by theoretical calculations, which correctly predicted the observed solvent dependence of the emission spectra. Similarly, the calculated IP and EA values were in excellent agreement with the experiment. DTP-TD, DTP-BTD, DTP-TZ, and DTP-NDI could be electropolymerized to yield polymers of very narrow electrochemical band gap and characterized by redox states differing in color coordinates and lightness. Poly(DTP-NDI) and poly(DTP-TD) showed promising electrochromic behavior, not only providing a rich color palette in the visible but also exhibiting near-infrared (NIR) electrochromism. © 2022 American Chemical Society.
Kulszewicz-Bajer I., Nowakowski R., Zagórska M., Maranda-Niedbała A., Mech W., Wróbel Z., Drapała J., Wielgus I., Korona K.P. Copolymers Containing 1-Methyl-2-phenyl-imidazole Moieties as Permanent Dipole Generating Units: Synthesis, Spectroscopic, Electrochemical, and Photovoltaic Properties
Molecules 2022, 27, 3, Art. No. 915
DOI: 10.3390/molecules27030915
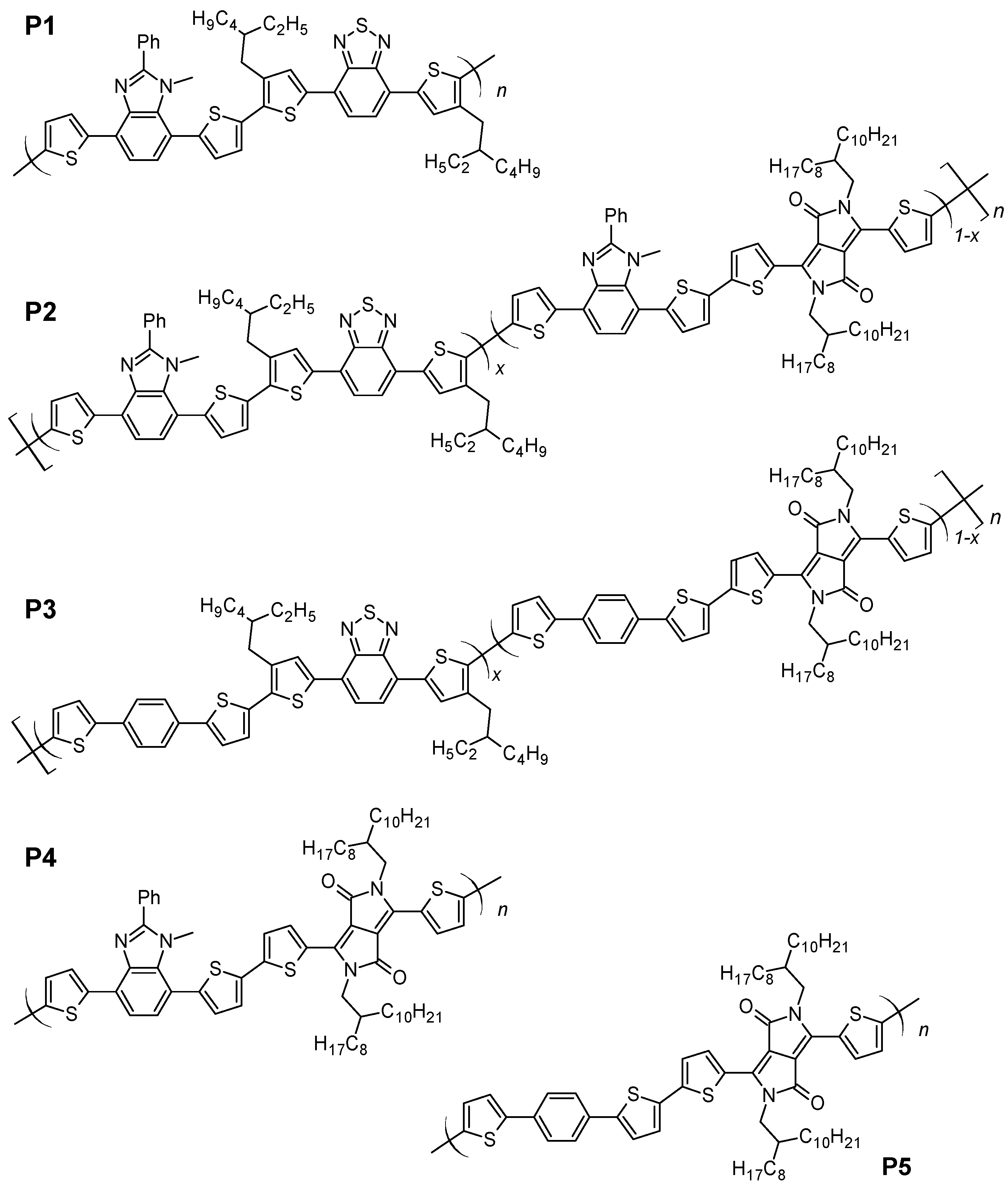
Abstract:New donor–acceptor conjugated alternating or random copolymers containing 1-methyl-2-phenylbenzimidazole and benzothiadiazole (P1), diketopyrrolopyrrole (P4), or both acceptors (P2) are reported. The specific feature of these copolymers is the presence of a permanent dipole-bearing moiety (1-methyl-2-phenyl imidazole (MPI)) fused with the 1,4-phenylene ring of the polymer main chain. For comparative reasons, polymers of the same main chain but deprived of the MPI group were prepared, namely, P5 with diketopyrrolopyrrole and P3 with both acceptors. The presence of the permanent dipole results in an increase of the optical band gap from 1.51 eV in P3 to 1.57 eV in P2 and from 1.49 eV in P5 to 1.55 eV in P4. It also has a measurable effect on the ionization potential (IP) and electrochemical band gap (EgCV), leading to their decrease from 5.00 and 1.83 eV in P3 to 4.92 and 1.79 eV in P2 as well as from 5.09 and 1.87 eV in P5 to 4.94 and 1.81 eV in P4. Moreover, the presence of permanent dipole lowers the exciton binding energy (Eb) from 0.32 eV in P3 to 0.22 eV in P2 and from 0.38 eV in P5 to 0.26 eV in P4. These dipole-induced changes in the polymer properties should be beneficial for photovoltaic applications. Bulk heterojunction solar cells fabricated from these polymers (with PC71BM acceptor) show low series resistance (rs), indicating good electrical transport properties. The measured power conversion efficiency (PCE) of 0.54% is limited by the unfavorable morphology of the active layer. © 2022 by the authors. Licensee MDPI, Basel, Switzerland.
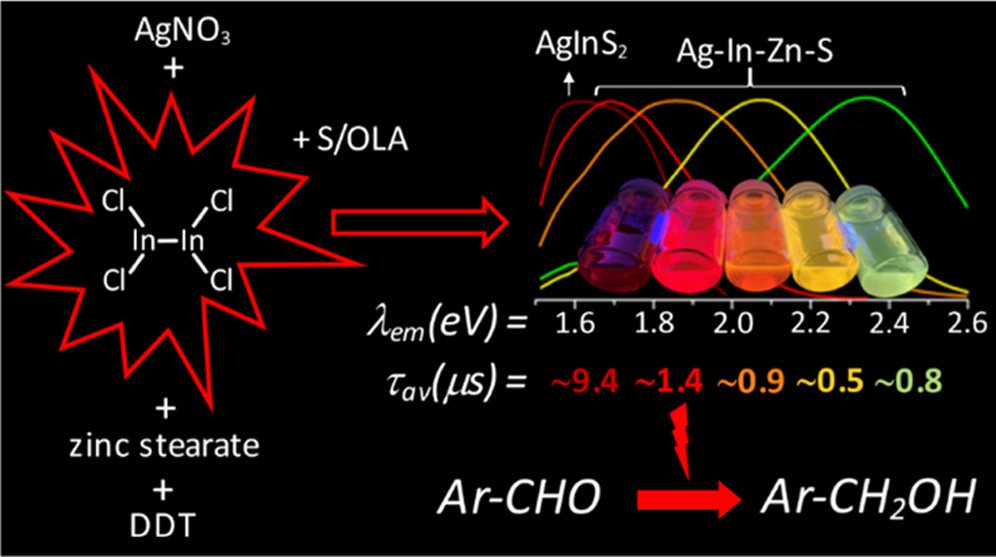
Kowalik P., Bujak P., Penkala M., Maroń A.M., Ostrowski A., Kmita A., Gajewska M., Lisowski W., Sobczak J.W., Pron A. Indium(II) Chloride as a Precursor in the Synthesis of Ternary (Ag-In-S) and Quaternary (Ag-In-Zn-S) Nanocrystals
Chemistry of Materials 2022, 34, 2, 809-825
DOI: 10.1021/acs.chemmater.1c03800
Abstract:A new indium precursor, namely, indium(II) chloride, was tested as a precursor in the synthesis of ternary Ag–In–S and quaternary Ag–In–Zn–S nanocrystals. This new precursor, being in fact a dimer of Cl2In–InCl2 chemical structure, is significantly more reactive than InCl3, typically used in the preparation of these types of nanocrystals. This was evidenced by carrying out comparative syntheses under the same reaction conditions using these two indium precursors in combination with the same silver (AgNO3) and zinc (zinc stearate) precursors. In particular, the use of indium(II) chloride in combination with low concentrations of the zinc precursor yielded spherical-shaped (D = 3.7–6.2 nm) Ag–In–Zn–S nanocrystals, whereas for higher concentrations of this precursor, rodlike nanoparticles (L = 9–10 nm) were obtained. In all cases, the resulting nanocrystals were enriched in indium (In/Ag = 1.5–10.3). Enhanced indium precursor conversion and formation of anisotropic, longitudinal nanoparticles were closely related to the presence of thiocarboxylic acid type of ligands in the reaction mixture. These ligands were generated in situ and subsequently bound to surfacial In(III) cations in the growing nanocrystals. The use of the new precursor of enhanced reactivity facilitated precise tuning of the photoluminescence color of the resulting nanocrystals in the spectral range from ca. 730 to 530 nm with photoluminescence quantum yield (PLQY) varying from 20 to 40%. The fabricated Ag–In–S and Ag–In–Zn–S nanocrystals exhibited the longest, reported to date, photoluminescence lifetimes of ∼9.4 and ∼1.4 μs, respectively. It was also demonstrated for the first time that ternary (Ag–In–S) and quaternary (Ag–In–Zn–S) nanocrystals could be applied as efficient photocatalysts, active under visible light (green) illumination, in the reaction of aldehydes reduction to alcohols. © 2022 The Authors. Published by American Chemical Society.
Kotwica K., Wielgus I., Proń A. Azaacenes based electroactive materials: Preparation, structure, electrochemistry, spectroscopy and applications-a critical review
Materials 2021, 14, 18, Art. No. 5155
DOI: 10.3390/ma14185155
Abstract:This short critical review is devoted to the synthesis and functionalization of various types of azaacenes, organic semiconducting compounds which can be considered as promising materials for the fabrication of n-channel or ambipolar field effect transistors (FETs), components of active layers in light emitting diodes (LEDs), components of organic memory devices and others. Emphasis is put on the diversity of azaacenes preparation methods and the possibility of tuning their redox and spectroscopic properties by changing the C/N ratio, modifying the nitrogen atoms distribution mode, functionalization with electroaccepting or electrodonating groups and changing their molecular shape. Processability, structural features and degradation pathways of these compounds are also discussed. A unique feature of this review concerns the listed redox potentials of all discussed compounds which were normalized vs. Fc/Fc+. This required, in frequent cases, recalculation of the originally reported data in which these potentials were determined against different types of reference electrodes. The same applied to all reported electron affinities (EAs). EA values calculated using different methods were recalculated by applying the method of Sworakowski and co-workers (Org. Electron. 2016, 33, 300–310) to yield, for the first time, a set of normalized data, which could be directly compared. © 2021 by the authors.
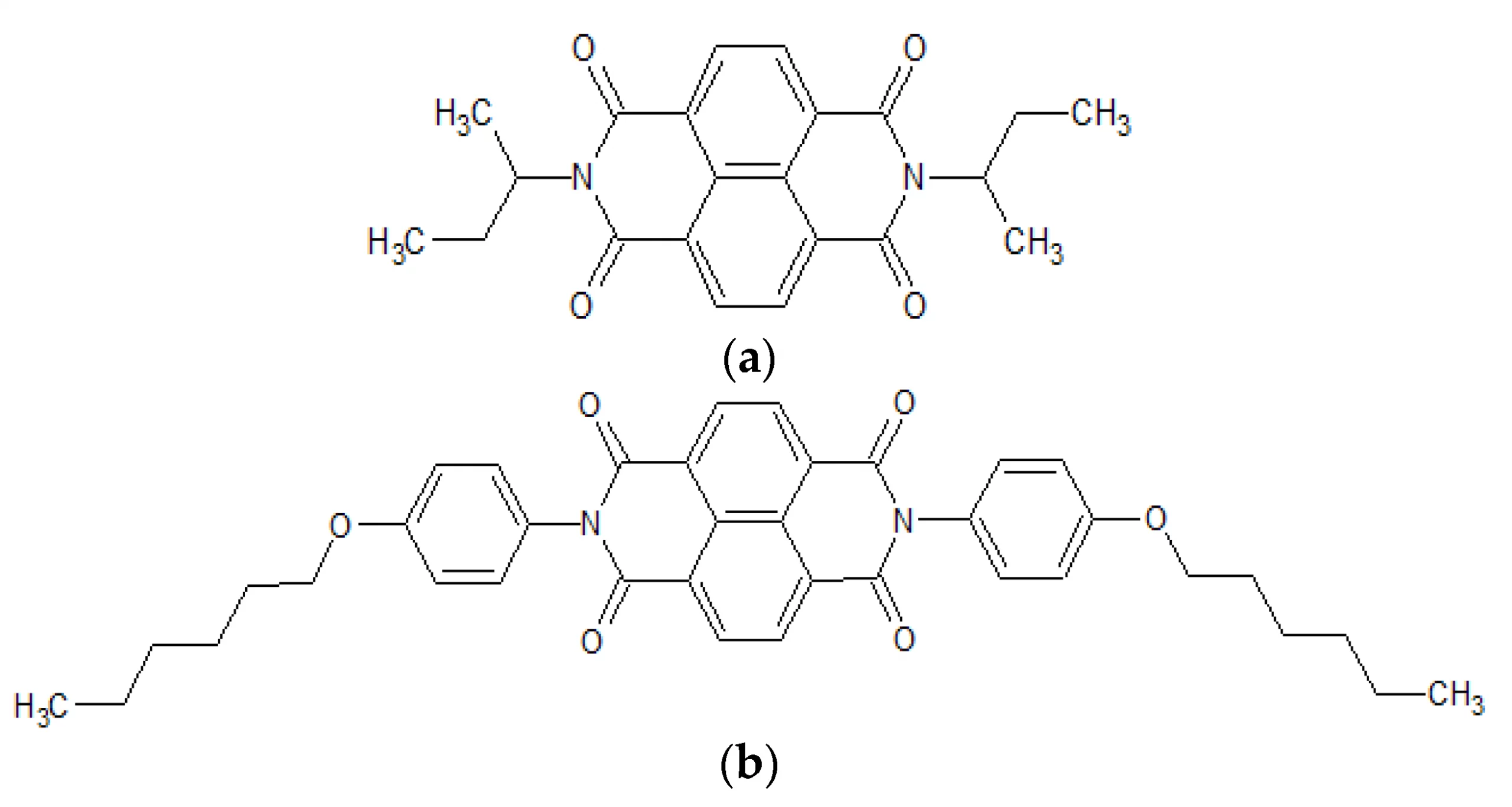
Jung J., Selerowicz A., Maczugowska P., Halagan K., Rybakiewicz-Sekita R., Zagorska M., Stefaniuk-Grams A. Electron transport in naphthalene diimide derivatives
Materials 2021, 14, 14, Art. No. 4026
DOI: 10.3390/ma14144026
Abstract: Two naphthalene diimides derivatives containing two different (alkyl and alkoxyphenyl) N-substituents were studied, namely, N,N′-bis(sec-butyl)-1,4,5,8-naphthalenetetracarboxylic acid diimide (NDI-s-Bu) and N,N′-bis(4-n-hexyloxyphenyl)-1,4,5,8-naphthalenetetracarboxylic acid diimide (NDI-4-n-OHePh). These compounds are known to exhibit electron transport due to their electron-deficient character evidenced by high electron affinity (EA) values, determined by electrochemical methods and a low-lying lowest unoccupied molecular orbital (LUMO) level, predicted by density functional theory (DFT) calculations. These parameters make the studied organic semiconductors stable in operating conditions and resistant to electron trapping, facilitating, in this manner, electron transport in thin solid layers. Current–voltage characteristics, obtained for the manufactured electron-only devices operating in the low voltage range, yielded mobilities of 4.3 × 10−4 cm2V−1s−1 and 4.6 × 10−6 cm2V−1s−1 for (NDI-s-Bu) and (NDI-4-n-OHePh), respectively. Their electron transport characteristics were described using the drift–diffusion model. The studied organic semiconductors can be considered as excellent candidates for the electron transporting layers in organic photovoltaic cells and light-emitting diodes © 2021 by the authors.
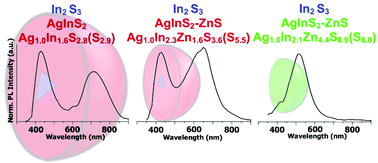
Kowalik P., Mucha S.G., Matczyszyn K., Bujak P., Mazur L.M., Ostrowski A., Kmita A., Gajewska M., Pron A. Heterogeneity induced dual luminescence properties of AgInS2and AgInS2-ZnS alloyed nanocrystals
Inorganic Chemistry Frontiers 2021, 8, 14, 3450-3462
DOI: 10.1039/d1qi00566a
Abstract: We have elaborated a new preparation method of ternary AgInS2 and alloyed quaternary AgInS2–ZnS nanocrystals which consisted of two consecutive injections of sulfur (S dissolved in oleylamine, OLA) and silver (AgNO3 or Ag2CO3 dissolved in dichlorobenzene, DCB) precursors into a mixture of indium(III) chloride, zinc stearate and 1-dodecanethiol (DDT) dissolved in 1-octadecene (ODE). Under these conditions, nucleation of cubic In2S3 seeds took place followed by the growth of orthorhombic AgInS2 or alloyed AgInS2–ZnS cubic phases to yield heterodimer type nanocrystals. In both types of nanocrystals clearly separated photoluminescence peaks could be observed, confirming their heterogenic nature. The first one at 430 nm originated from the luminescence of the In2S3 phase. The second one was ascribed to the presence of a ternary Ag–In–S or quaternary Ag–In–Zn–S phase and its position, within the spectroscopic range from 515 nm to 710 nm, strongly dependent on the nanocrystal composition. The registered two-dimensional excitation–emission topographical maps clearly indicated that the observed emissions in two different spectral regions were related to the excitation in the same spectral range (300–400 nm); however their photoluminescence mechanisms were distinctly different. The photoluminescence lifetime of ∼3 ns measured for the emission at shorter wavelengths was typical of the simple mechanism of exciton radiative recombination in the In2S3 phase. The significantly longer lifetime of the longer wavelength emission (∼26 μs) seemed to clearly indicate that in this case the photoluminescence mechanism was more complex, involving exciton trap states whose positions depended on the composition of the ternary (or quaternary) phase. © the Partner Organisations.
Kowalik P., Bujak P., Penkala M., Pron A. Organic-to-aqueous phase transfer of alloyed AgInS2-ZnS nanocrystals using simple hydrophilic ligands: Comparison of 11-mercaptoundecanoic acid, dihydrolipoic acid and cysteine
Nanomaterials 2021, 11, 4, Art. No. 843
DOI: 10.3390/nano11040843
Abstract: The exchange of primary hydrophobic ligands for hydrophilic ones was studied for two types of alloyed AgInS2-ZnS nanocrystals differing in composition and by consequence exhibiting two different emission colors: red (R) and green (G). Three simple hydrophilic ligands were tested, namely, 11-mercaptoundecanoic acid, dihydrolipoic acid and cysteine. In all cases, stable aqueous colloidal dispersions were obtained. Detailed characterization of the nanocrystal surface before and after the ligand exchange by NMR spectroscopy unequivocally showed that the exchange process was the most efficient in the case of dihydrolipoic acid, leading to the complete removal of the primary ligands with a relatively small photoluminescence quantum yield drop from 68% to 40% for nanocrystals of the R type and from 48% to 28% for the G ones. © 2021 by the authors. Licensee MDPI, Basel, Switzerland.
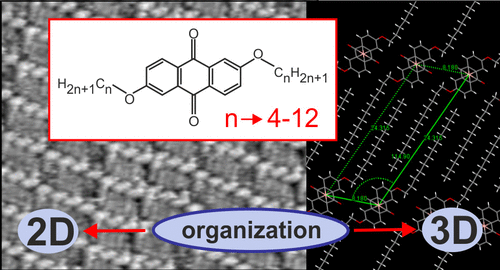
Maranda-Niedbała A., Krzyżewska K., Kotwica K., Skórka L., Drapała J., Jarzembska K.N., Zagórska M., Proń A., Nowakowski R. 9,10-Anthraquinones Disubstituted with Linear Alkoxy Groups: Spectroscopy, Electrochemistry, and Peculiarities of Their 2D and 3D Supramolecular Organizations
Langmuir 2020, 36, 49, 15048-15063
DOI: 10.1021/acs.langmuir.0c02586
Abstract:Spectroscopic, electrochemical, and structural properties of 2,6-dialkoxy-9,10-anthraquinones (Anth-OCn, n = 4, 6, 8, 10, and 12) of increasing alkoxy substituents length were investigated. UV–vis spectroscopy showed a substitution-induced bathochromic shift of the least energetic band from 325 nm in the case of unsubstituted anthraquinone to ca. 350 nm for the studied derivatives. Similarly as unsubstituted anthraquinone, the studied compound showed two reversible one electron reductions to a radical anion and spinless anions, respectively. The first reduction was affected by electron-donating properties of the substituents, its potential being shifted to ca. −1.5 V (vs Fc/Fc+), i.e., by 80 to 95 mV as compared to the case of unsubstituted anthraquinone. This corresponded to a decrease of |EA| from 3.27 to 3.19–3.17 eV. The experimental spectroscopic and electrochemical data were in full agreement with the DFT calculations. The introduction of the alkoxy substituent improved solution processibility of the studied compounds and facilitated the formation of their ordered supramolecular 2D aggregation on HOPG as well as single crystal growth from solutions. Comparative structural investigations carried out on single crystals and monolayers deposited on HOPG revealed two, mutually related, effects of the substituent length on the resulting supramolecular organization. The first one concerns both the 2D organization in the monolayers and 3D molecular arrangement in crystals: increasing substituent length evolution of the structure occurs from herringbone-type to lamellar. The second effect, observed in monolayers of the derivatives with longer substituents, concerns gradual evolution of their lamellar structures with increasing substituent length. This evolution is induced by the structure of the graphite substrate and involves increasing correlation of the molecules orientation (anthraquinone cores as well as alkoxy substituents) with the symmetry of the graphite substrate. As a result, their 2D and 3D structures become dissimilar. © 2020 American Chemical Society.
Rybakiewicz R., Ganczarczyk R., Charyton M., Skorka L., Ledwon P., Nowakowski R., Zagorska M., Pron A. Low band gap donor-acceptor-donor compounds containing carbazole and naphthalene diimide units: Synthesis, electropolymerization and spectroelectrochemical behaviour
Electrochimica Acta 2020, 358, Art. No. 136922
DOI: 10.1016/j.electacta.2020.136922
Abstract:A new DπAπD compound consisting of naphthalene diimide core-functionalized with 1,4-phenylene-functionalized carbazole, namely 2,6-bis[4-(9H-carbazol-9-yl)phenyl]-N,N’-bis(2-ethylhexyl)-1,4,5,8-naphthalenetetracarboxylic acid diimide (CNDI) was synthesized. Its redox properties (reduction and oxidation potentials, electron affinities (EA) and ionization potentials (IP)) were compared with those of 4,7-bis(4-(9H-carbazol-9-yl)phenyl)benzo[c][1,2,5]thiadiazole (CBTD) – a specially synthesized reference compound containing identical Dπ- units and significantly weaker acceptor (benzothiadiazole). It was demonstrated that the acceptor strength, determined as the index of the spatial extent of the charge transfer, DCTP, only weakly influenced the molecule oxidation potential and by consequence its ionization potential. To the contrary, strong effect of charge transfer, expressed by DCTP, was found for the reduction potential of CNDI and CBTD, showing significantly higher reduction potential of the former leading to much higher |EA|. CNDI could be electrochemically polymerized yielding a low band gap polymer (Eg = 1.63 eV) of ambipolar character, exhibiting low ionization potential (IP = 5.48 eV) and high electron affinity (|EA| = 3.85 eV). In its cyclic voltammograms polyCNDI shows five redox states, namely neutral, first and second reduction and first and second oxidation ones. As revealed by detailed studies of electrochromic parameters only three redox states (neutral, first reduction and first oxidation) demonstrate good electrochromic performance. © 2020
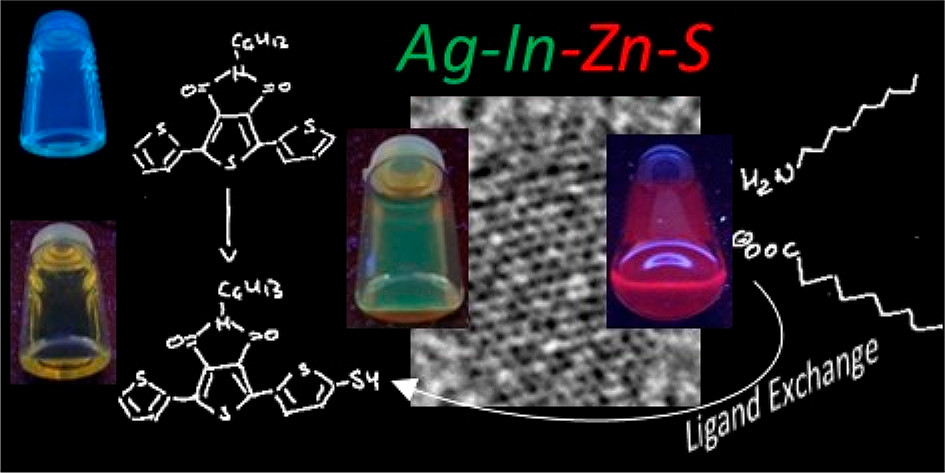
Kowalik P., Bujak P., Wróbel Z., Penkala M., Kotwica K., Maroń A., Pron A. From Red to Green Luminescence via Surface Functionalization. Effect of 2-(5-Mercaptothien-2-yl)-8-(thien-2-yl)-5-hexylthieno[3,4- c]pyrrole-4,6-dione Ligands on the Photoluminescence of Alloyed Ag-In-Zn-S Nanocrystals
Inorganic Chemistry 2020, 59, 19, 14594-14604
DOI: 10.1021/acs.inorgchem.0c02468
Abstract: A semiconducting molecule containing a thiol anchor group, namely 2-(5-mercaptothien-2-yl)-8-(thien-2-yl)-5-hexylthieno[3,4-c]pyrrole-4,6-dione (abbreviated as D-A-D-SH), was designed, synthesized, and used as a ligand in nonstoichiometric quaternary nanocrystals of composition Ag1.0In3.1Zn1.0S4.0(S6.1) to give an inorganic/organic hybrid. Detailed NMR studies indicate that D-A-D-SH ligands are present in two coordination spheres in the organic part of the hybrid: (i) inner in which the ligand molecules form direct bonds with the nanocrystal surface and (ii) outer in which the ligand molecules do not form direct bonds with the inorganic core. Exchange of the initial ligands (stearic acid and 1-aminooctadecane) for D-A-D-SH induces a distinct change of the photoluminescence. Efficient red luminescence of nanocrystals capped with initial ligands (λmax = 720 nm, quantum yield = 67%) is totally quenched and green luminescence characteristic of the ligand appears (λmax = 508 nm, quantum yield = 10%). This change of the photoluminescence mechanism can be clarified by a combination of electrochemical and spectroscopic investigations. It can be demonstrated by cyclic voltammetry that new states appear in the hybrid as a consequence of D-A-D-SH binding to the nanocrystals surface. These states are located below the nanocrystal LUMO and above its HOMO, respectively. They are concurrent to deeper donor and acceptor states governing the red luminescence. As a result, energy transfer from the nanocrystal HOMO and LUMO levels to the ligand states takes place, leading to effective quenching of the red luminescence and appearance of the green one. © 2020 American Chemical Society.
Rybakiewicz R., Ganczarczyk R., Wiosna-Salyga G., Bobowska I., Banasiewicz M., Charyton M., Skorka L., Zagorska M., Pron A. Luminescent organic materials based on donor-acceptor-donor compounds containing carbazole donors and acceptors of varying strength: Spectroscopy, redox properties and application in organic light emitting diodes
Optical Materials 2020, 108, Art. No. 110428
DOI: 10.1016/j.optmat.2020.110428
Abstract:Three D-π-Α-π-D organic luminophores of tunable donor-acceptor interactions were synthesized by combining carbazole D-units with two different A units, namely 2,5-dithiophen-2-yl-[1,3,4]-thiadiazole and benzo[c][1,2,5]thiadiazole. In the third compound tert-butyl groups were introduced to the carbazole ring to increase its electron-donating properties. This compound, namely 4,7-bis(4-(3,6-di-tert-butyl-9H-carbazol-9-yl)phenyl)benzo[c][1,2,5]thiadiazole (CBTD2) turned out to be a very good photoluminophore showing the photoluminescence quantum yield (φ) of 62% in toluene solution and 45% when dispersed in a solid blend consisting of poly(vinylcarbazole) (PVK) and 2-([1,1′-biphenyl]-4-yl)-5-(4′-(tert-butyl)-[1,1′-phenyl]-4-yl)-1,3,4-oxadiazole (PBD). Moreover, its electrochemically determined ionization potential (IP = 5.55 eV) and electron affinity (EA = −2.73 eV) made it a very promising candidate for application as an active component in light emitting diodes of guest/host configuration with PVK-PBD matrix. The fabricated diodes of the following structure ITO/PEDOT:PSS/PVK-PBD + CBTD2/Ca/Ag showed the luminance reaching 6000 cd/m2 with luminous efficiency of 1 cd/A. © 2020 Elsevier B.V.
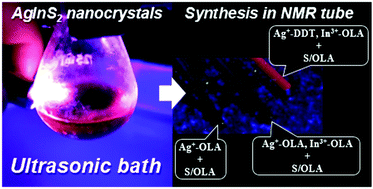
Kowalik P., Penkala M., Bujak P., Kmita A., Gajewska M., Ostrowski A., Slodek A., Pron A. From Ag2S to luminescent Ag-In-S nanocrystals: Via an ultrasonic method-an in situ synthesis study in an NMR tube
Journal of Materials Chemistry C 2020, 8, 26, 8942-4952
DOI: 10.1039/d0tc01880e
Abstract: Starting from simple metal precursors (AgNO3 and InCl3), ligands (oleylamine (OLA) and 1-dodecanethiol (DDT)) and popular sulfur precursor S/OLA, we have developed a new method for the preparation of colloidal Ag2S nanocrystals and highly luminescent ternary AgInS2 nanocrystals. The significant advantage of this method is that the nanocrystals can be prepared at room temperature in an ultrasonic bath, without the need for an inert gas atmosphere. In particular, using this method, it was possible to perform the synthesis of the studied nanocrystals in an NMR tube providing direct analytical access to chemical transformations occurring in the reaction mixture during the nanocrystal synthesis. Detailed analysis of the evolution of NMR signals originating from ligands (OLA and DDT) enabled us to identify the nucleation and growth stages as well as the stabilization stage of the nanocrystal formation i.e. the stage in which the ligands were permanently attached to the nanocrystal surface. We also demonstrated that under the analyzed conditions, the reactivities of silver and indium precursors resulting in appropriate cation-to-ligand connections (Ag+-DDT and In3+-OLA) were crucial for the nucleation of ternary AgInS2 nanocrystals exhibiting intense red luminescence. This journal is © The Royal Society of Chemistry.
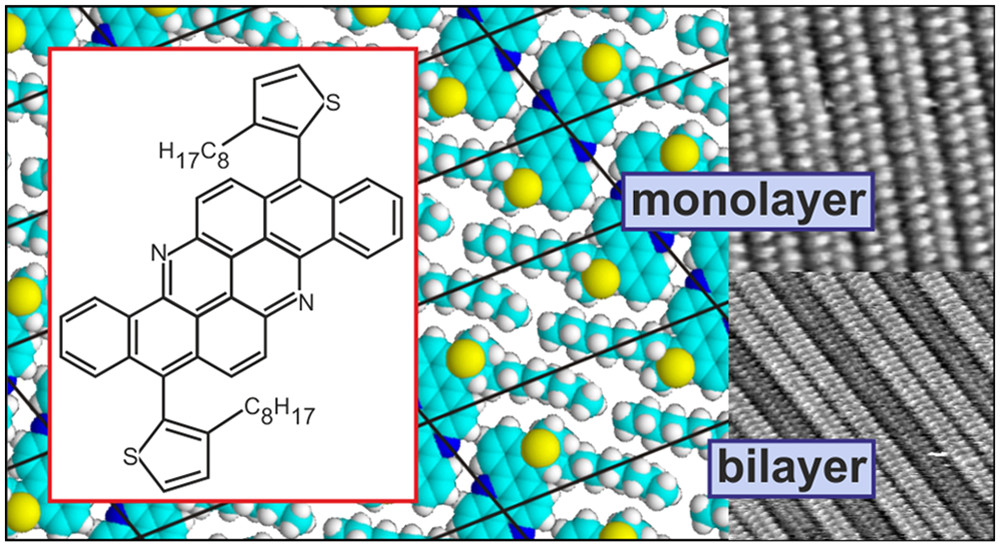
Jaroch T., Maranda-Niedbała A., Krzyżewska K., Kotwica K., Bujak P., Skórka L., Zagórska M., Proń A., Nowakowski R. Self-Assembly Properties of Solution Processable, Electroactive Alkoxy, and Alkylthienylene Derivatives of Fused Benzoacridines: A Scanning Tunneling Microscopy Study
Langmuir 2020, 36, 19, 5417-5427
DOI: 10.1021/acs.langmuir.9b03966
Abstract: Self-organization in mono- and bilayers on HOPG of two groups of benz[5,6]acridino[2,1,9,8-klmna]acridine derivatives, namely, 8,16-dialkoxybenzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridines with an increasing alkoxy substituent length and 8,16-bis(3- or 4- or 5-octylthiophen-2-yl)benzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridines, i.e., three positional isomers of the same benzoacridine, is investigated by scanning tunneling microscopy. The layers were deposited from a solution of the adsorbate (in hexane or dichloromethane) and imaged ex situ at molecular resolution. In all cases, the resulting two-dimensional (2D) supramolecular organization is governed by the interactions between large, fused heteroaromatic cores that form densely packed rows separated by areas covered by substituents. In 8,16-dialkoxybenzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridines, the alkoxy substituents, separating the rows of densely packed cores, are interdigitated. An increasing substituent length leads to an intuitively expected increase in this 2D unit cell parameter that corresponds to the orientation of the substituent in the monolayer. In the case of 8,16-bis(3- or 4- or 5-octylthiophen-2-yl)benzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridine positional isomers, the self-assembly processes are more complex. Although the determined 2D unit cell is in all cases essentially the same, the role of alkylthienylene substituents in layer formation is distinctly different. Thus, the formation of monolayers and bilayers is very sensitive to isomerism. 8,16-Bis(5-octylthiophen-2-yl)benzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridine is capable of forming the most stable monolayer and the most labile bilayer. In the case of 8,16-bis(3-octylthiophen-2-yl)benzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridine, an inverse phenomenon is observed leading to the most labile monolayer and the most stable bilayer. These differences are rationalized in terms of dissimilar molecular geometries of the studied isomers and different interdigitation patterns in their 2D supramolecular structures. Copyright © 2020 American Chemical Society.
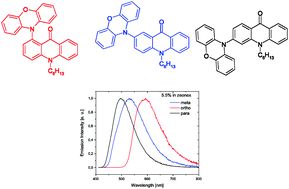
Kulszewicz-Bajer I., Zagorska M., Banasiewicz M., Guńka P.A., Toman P., Kozankiewicz B., Wiosna-Salyga G., Pron A. Effect of the substituent position on the electrochemical, optical and structural properties of donor-acceptor type acridone derivatives
Physical Chemistry Chemical Physics 2020, 22, 16, 8522-8534
DOI: 10.1039/d0cp00521e
Abstract: Three new donor-acceptor (D-A) compounds, positional isomers of phenoxazine-substituted acridone, namely 1-phenoxazine-N-hexylacridone ( o-A ), 2-phenoxazine-N-hexylacridone ( m-A ) and 3-phenoxazine-N-hexylacridone ( p-A ), were synthesized. The synthesized compounds showed interesting, isomerism-dependent electrochemistry. Their oxidation was reversible and their potential (givenvs.Fc/Fc+) changed from 0.21 V for o-A to 0.36 V for p-A . In contrast, their reduction was irreversible, isomerism-independent and occurred at rather low potentials (ca.−2.25 to −2.28 V). The electrochemical results led to the following values of the ionization potentials (IPs) and electron affinities (EAs): 5.03 eV and −2.14 eV, 5.15 eV and −2.20 eV, and 5.20 eV and −2.28 eV for o-A m-A and p-A , respectively. The experimentally obtained values were in very good agreement with those predicted by DFT calculations. All three isomers readily formed single crystals suitable for their structure determination. o-A and p-A crystallized inP1̄ andP21/nspace groups, respectively, with one molecule per asymmetric unit, while m-A crystallized in theP21/cspace group with two molecules in the asymmetric unit accompanied by disordered solvent molecules. The UV-vis spectra of the studied compounds were isomerism and solvent independent, yielding absorption maxima in the vicinity of 400 nm. Their photoluminescence spectra, in turn, strongly depended on isomerism and the used solvent showing smaller Stokes shifts for the emission bands registered in toluene as compared to the corresponding bands measured in dichloromethane. The photoluminescence quantum yields (ϕ) were systematically higher for toluene solutions reaching the highest value of 20% for p-A . For all three isomers studied, stationary and time-resolved spectroscopic investigations carried out in toluene at different temperatures revealed spectral features indicating a contribution of thermally activated delayed fluorescence (TADF) to the observed spectroscopic behaviour. The measured photoluminescence quantum yields (ϕ) were higher for solid state films of pure compounds and for their dispersions in solid matrices (zeonex) than those recorded for toluene and dichloromethane solutions of the studied phenoxazine-N-hexylacridone isomers. The obtained experimental spectroscopic and structural data were confronted with theoretical predictions based on DFT calculations. © the Owner Societies 2020.
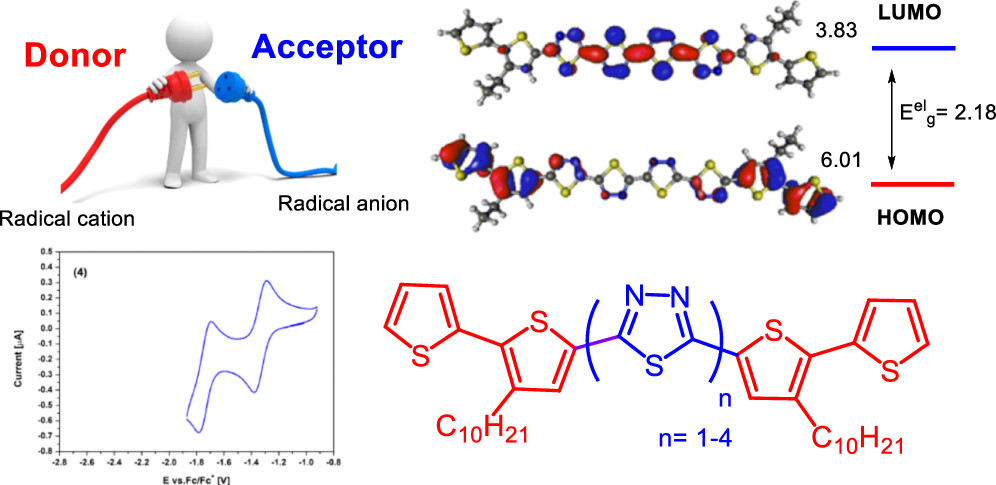
Kostyuchenko A.S., Kurowska A., Zassowski P., Zheleznova T.Y., Ulyankin E.B., Domagala W., Pron A., Fisyuk A.S. Synthesis of Bis([2,2′-bithiophen]-5-yl)-Substituted Oligothiadiazoles: Effect of the Number of Acceptor Units on Electrochemical and Spectroscopic Properties
Journal of Organic Chemistry 2019, 84, 16, 10040-10049
DOI: 10.1021/acs.joc.9b01216
Abstract: 1,3,4-Thiadiazole, 2,2′-bi(1,3,4-thiadiazole), 2,2′:5′,2″-ter(1,3,4-thiadiazole), and 2,2′:5′,2″:5″,2′-quater(1,3,4-thiadiazole) symmetrically disubstituted with 3-alkyl-(2,2′-bithiophen)-5-yl were synthesized by new procedures using readily available ethyl 3-alkyl-(2,2′-bithiophene)-5-carboxylate as a convenient substrate. These new compounds with a fixed number of donor rings and increasing number of acceptor rings showed very interesting, tunable redox properties. In particular, they exhibited electron affinities (EAs) ranging from -3.06 to -3.83 eV, reaching EA values desired for air-operating n-type organic semiconductors. Their electrochemically determined ionization potentials were only moderately dependent on the number of thiadiazole rings, varying from 5.83 to 6.01 eV. Emission spectra of these compounds could also be tuned in a wide range (from 470 to 600 nm). Spectroscopic and electrochemical data were confirmed by density functional theory calculations demonstrating full consistency. © 2019 American Chemical Society.
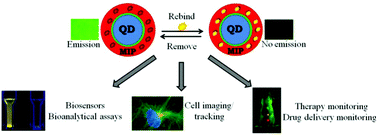
Sobiech M., Bujak P., Luliński P., Pron A. Semiconductor nanocrystal-polymer hybrid nanomaterials and their application in molecular imprinting
Nanoscale 2019, 11, 25, 12030-12074
DOI: 10.1039/c9nr02585e
Abstract: Quantum dots (QDs) are attractive semiconductor fluorescent nanomaterials with remarkable optical and electrical properties. The broad absorption spectra and high stability of QD transducers are advantageous for sensing and bioimaging. Molecular imprinting is a technique for manufacturing synthetic polymeric materials with a high recognition ability towards a target analyte. The high selectivity of the molecularly imprinted polymers (MIPs) is a result of the fabrication process based on the template-tailored polymerization of functional monomers. The three-dimensional cavities formed in the polymer network can serve as the recognition elements of sensors because of their specificity and stability. Appending specific molecularly imprinted layers to QDs is a promising strategy to enhance the stability, sensitivity, and selective fluorescence response of the resulting sensors. By merging the benefits of MIPs and QDs, inventive optical sensors are constructed. In this review, the recent synthetic strategies used for the fabrication of QD nanocrystals emphasizing various approaches to effective functionalization in aqueous environments are discussed followed by a detailed presentation of current advances in QD conjugated MIPs (MIP-QDs). Frontiers in manufacturing of specific imprinted layers of these nanomaterials are presented and factors affecting the specific behaviour of an MIP shell are identified. Finally, current limitations of MIP-QDs are defined and prospects are outlined to amplify the capability of MIP-QDs in future sensing. © 2019 The Royal Society of Chemistry.
Pron A., Bujak P., Zagorska M., Cho N., Kim T.-D., Lee K.-S. Synthesis of solution-processable nanoparticles of inorganic semiconductors and their application to the fabrication of hybrid materials for organic electronics and photonics
Solution-Processable Components for Organic Electronic Devices 2019, pages 57-180
DOI: 10.1002/9783527813872.ch2
Abstract: Colloidal semiconductor nanocrystals consist of an inorganic core determining their physical properties and a shell of organic ligands that induce their colloidal stability in a wide range of solvents, both nonpolar and polar. They are considered as extremely promising nanomaterials for modern electronics. The band gaps of these nanocrystalline semiconductors can be conveniently tuned by their composition, type of crystal structure, and size (via quantum confinement), yielding nanomaterials whose physical and photophysical properties are unmatched by conventional materials. In the first part of the chapter, binary chalcogenide-type nanocrystals are described (CdS, CdSe, and CdTe) with special emphasis on their synthesis, photoluminescence quantum yield (PLQY) improvement through the preparation of core/shell systems, and/or alloying. The effect of organic ligands binding to the nanocrystal surface is described in detail because they not only assure the colloidal stability of these nano-objects but also influence to a certain extent their physicochemical properties. Ligand exchange procedures are discussed, which render the nanocrystals hydrophilic. The role of linker ligands is described, which assures further surface functionalization through grafting molecules (or macromolecules) of interest to the nanocrystal surface. The synthesis and functionalization of nanocrystals that do not contain toxic elements are illustrated in the subsequent part of the chapter, among them ternary Cu(Ag)-In-S(Se) and quaternary Cu(Ag)-In-Zn-S(Se), Cu-Zn-Sn-S(Se) nanocrystals, which can be considered not only as alternatives to binary nanocrystals but also significantly broaden their application spectrum. In addition to these indepth study of nanocrystals chemistry, we introduced the possible application of photopatternable nanocrystals in the microfabrication of 2D/3D functional structures. Energy and charge transfer in the semiconducting nanocrystal based organic-inorganic hybrid materials are also described. Lastly, we discussed recent development of metal halide perovskite materials and their applications in the field of optoelectronics. Recently developed low-dimensional perovskite materials are introduced with their light emitting device applications. © 2019 Wiley-VCH Verlag GmbH & Co. KGaA.
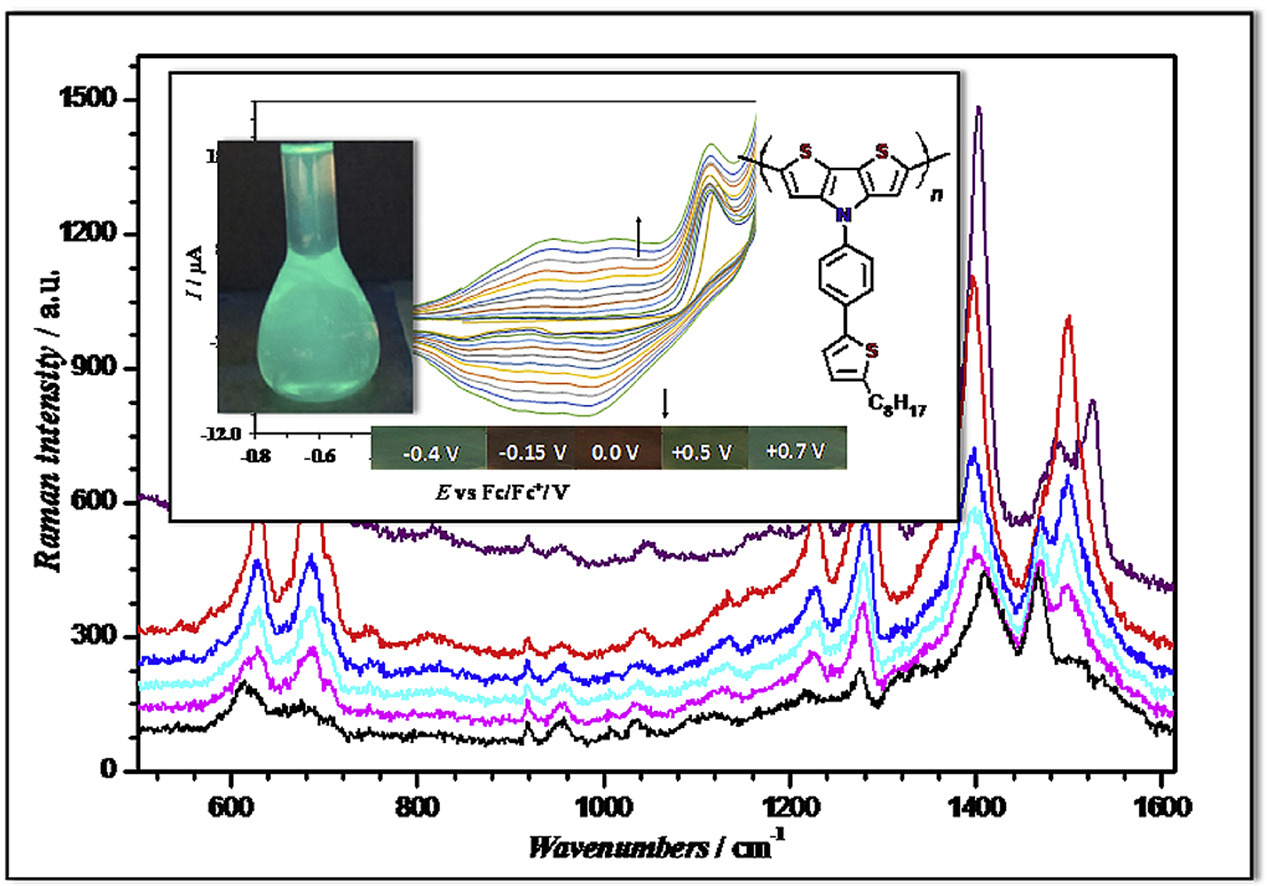
Rybakiewicz R., Skorka L., Louarn G., Ganczarczyk R., Zagorska M., Pron A. N-substituted dithienopyrroles as electrochemically active monomers: Synthesis, electropolymerization and spectroelectrochemistry of the polymerization products
Electrochimica Acta 2019, 295, 452-483
DOI: 10.1016/j.electacta.2018.10.123
Abstract: Two new electropolymerizable monomers were synthesized, namely dithieno[3,2-b:2′,3′-d]pyrrole N-functionalized with 4-(2-heptylthiazol-4-yl)phenyl (DTP1) and 4-(5-octylthiophen-2-yl)phenyl (DTP2) groups. Both monomers readily electropolymerize to yield the corresponding polyDTP1 and polyDTP2. Electrochemically determined ionization potential (IP) of both polymers (about 4.75 eV) are higher than IPs of poly(dithieno[3,2-b:2′,3′-d]pyrrole) N-substituted with alkyl or alkylphenyl groups. This finding, corroborated by DFT calculations, suggests that electron accepting nature of (thiazol-4-yl)phenyl and (thiophen-2-yl)phenyl substituents lowers the π-electron density in the dithienopyrrole moiety making the polymers oxidation more difficult. Optical band gaps of polyDTP1 (Eg opt = 1.76 eV) and polyDTP2 (Eg opt = 1.78 eV) are lower than the gaps of poly(dithieno[3,2-b:2′,3′-d]pyrrole) N-substituted with alkyl or alkylphenyl groups. This combined with higher IP value yield higher electron affinity (ǀEAǀ) value. Thus, the obtained new polymers are more difficult to oxidize but easier to reduce as compared to poly(dithieno[3,2-b:2′,3′-d]pyrrole)s studied to date. Again, these findings are in a very good agreement with DFT calculations. As evidenced by UV–vis–NIR spectroelectrochemistry, both polymers undergo classical (for conjugated polymers) oxidation, involving the formation of polarons in the first step and bipolarons in the second one. An interesting feature of the oxidation of polyDTP2 is the highly delocalized nature of bipolarons, indicative of the metallic state (featureless absorption tails extending towards NIR part of the spectrum). In order to elucidate the exact nature of the electrochemical oxidation process detailed Raman spectroelectrochemical investigations of polyDTP2 were carried out supported by the vibrational model calculations using two methods: DFT and General Valence Force Field (GVFF). This combined experimental and theoretical study leads to a conclusion that radical cations (polarons) formed at the first stage of oxidation are formed in the pyrrole ring whereas dications formed in the second stage show classical bipolaron configuration with positive charges located on the thiophene rings. © 2018
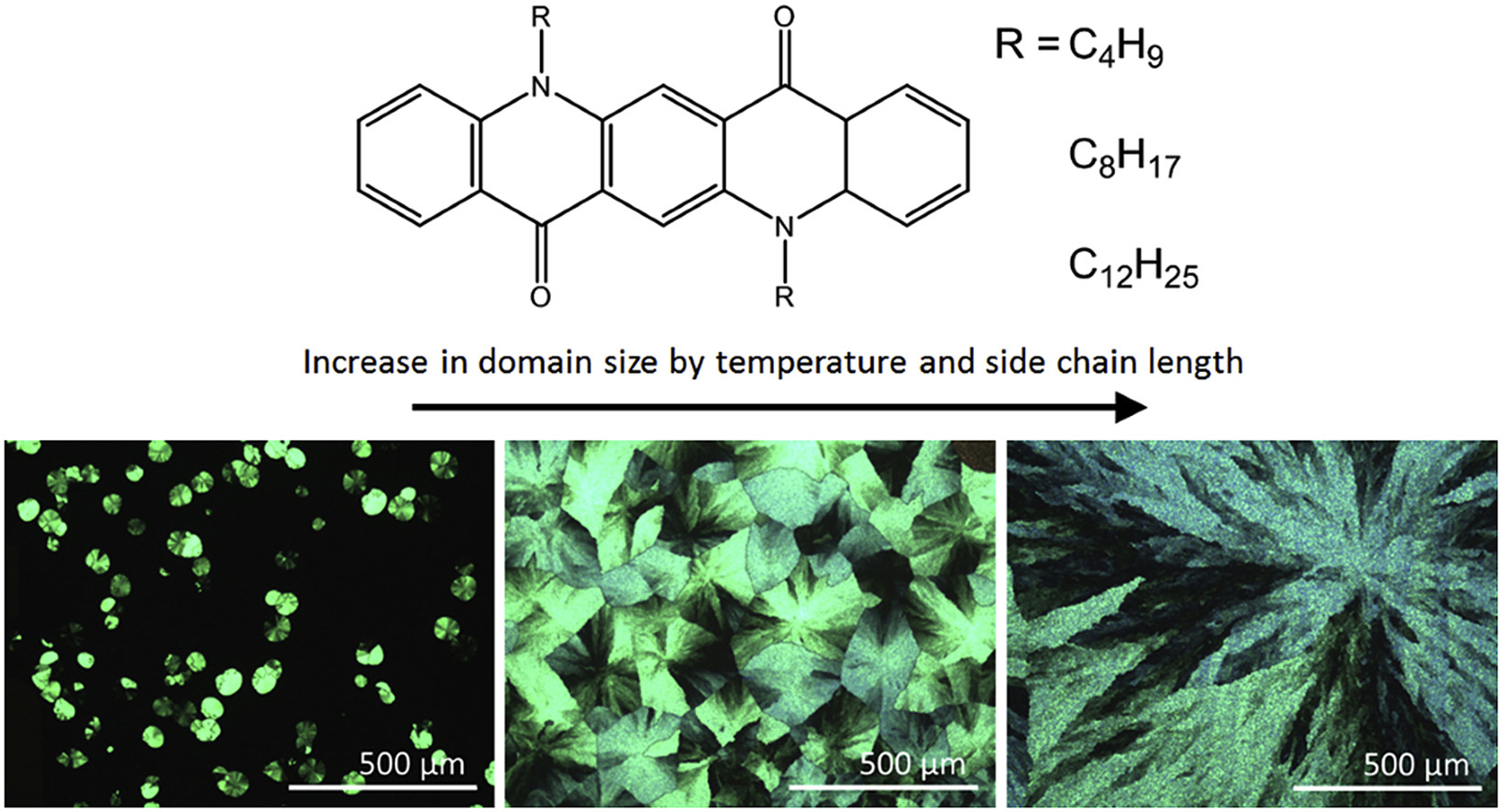
Marszalek T., Krygier I., Pron A., Wrobel Z., Blom P.M.W., Kulszewicz-Bajer I., Pisula W. Self-assembly and charge carrier transport of sublimated dialkyl substituted quinacridones
Organic Electronics 2019, 65, 127-134
DOI: 10.1016/j.orgel.2018.11.004
Abstract: Quinacridone, an industrial pigment, has recently shown a high charge carriers mobility in field-effect transistors. In search for new cheap organic semiconductors of improved vacuum processability we have synthesized three dialkyl derivatives of quinacridone, namely N,N′-dialkylquinacridones (alkyl = butyl, octyl, dodecyl), abbreviated as QA-C4, QA-C8 and QA-C12. The alkylation of quinacridone results in a significant decrease of its melting temperature which drops from 390 °C for quinacridone to 261 °C, 177 °C and 134 °C for QA-C4, QA-C8 and QA-C12, respectively, while retaining the onset of thermal decomposition above 390 °C. The elimination of the hydrogen bonding network between the carbonyl groups and amine hydrogens through alkylation not only lowers the melting temperature, but also induces supramolecular ordering in contrast to unsubstituted quinacridone. Detailed morphological and structural investigations of the vacuum deposited thin films have revealed that the length of the alkyl substituent is crucial for the molecular self-organization. Compound QA-C4 forms poorly ordered films, whereas QA-C8 and QA-C12 grow into a spherulitic dense morphology with increasing domain size at higher deposition temperatures. The more pronounced morphology is related to the lower melting point of the compounds and strong molecular diffusion during deposition. The poorly ordered films of QA-C4 do not show any field-effect response, what is consistent with previous reports. In contrast, transistors with QA-C8 or QA-C12 as active layers exhibit hole transport. Optimization of the deposition temperature, in which nucleation and crystal growth are properly balanced, resulted in OA-C8-based transistors with a hole mobility of 0.3 cm2/V, i.e. higher than in devices with unsubstituted quinacridone. © 2018 Elsevier B.V.
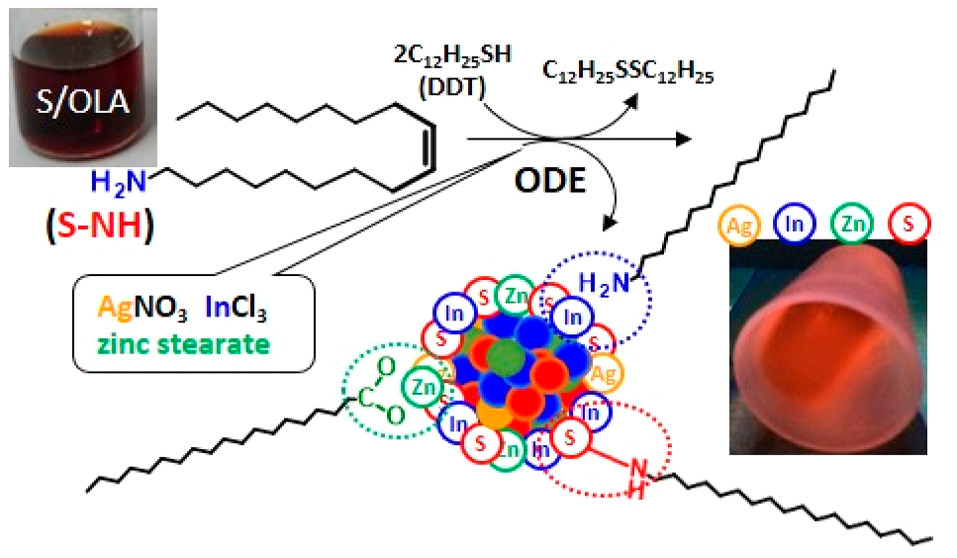
Bujak P., Wróbel Z., Penkala M., Kotwica K., Kmita A., Gajewska M., Ostrowski A., Kowalik P., Pron A. Highly Luminescent Ag-In-Zn-S Quaternary Nanocrystals: Growth Mechanism and Surface Chemistry Elucidation
Inorganic Chemistry 2019, 58, 2, 1358-1370
DOI: 10.1021/acs.inorgchem.8b02916
Abstract: The presented research is focused on the synthesis of alloyed Ag-In-Zn-S colloidal nanocrystals from a mixture of simple metal precursors such as AgNO 3 , InCl 3 , zinc stearate combined with 1-dodecanethiol (DDT), 1-octadecene (ODE), and sulfur dissolved in oleylamine (OLA). In particular, the focus is on the effect of the solvent (ODE vs 1,2-dichlorobenzene (DCB)) and the type of sulfur precursor (S/OLA vs S/n-octylamine (OCA)) on the metal precursors reactivates and on the chemical composition, crystal structure, and luminescent properties of the resulting nanocrystals. The replacement of ODE by DCB as a solvent lowers the reactivity of metal precursors and results in a 3-fold decrease of the photoluminescence quantum yields (Q.Y.) values (from 67% to 21%). This negative effect can be fully compensated by the use of S/OCA as a source of sulfur instead of S/OLA (Q.Y. increases from 21% to 64%). NMR studies of the isolated organic phase indicate that the S/OLA precursor generates two types of ligands being products of (Z)-1-amino-9-octadecene (OLA) hydrogenation. These are „surface bound” 1-aminooctadecane (C 18 H 37 NH 2 ) and crystal bound, i.e., alkyl chain covalently bound to the nanocrystal surface via surfacial sulfur (C 18 H 37 -NH-S crystal). Highly luminescent Ag-In-Zn-S nanocrystals exhibit a cation-enriched (predominantly indium) surface and are stabilized by a 1-aminooctadecane ligand, which shows more flexibility than OLA. These investigations were completed by hydrophilization of nanocrystals obtained via exchange of the primary ligands for 11-mercaptoundecanoic acid, (MUA) with only a 2-fold decrease of photoluminescence Q.Y. in the most successful case (from 67% to 31%). Finally, through ligand exchange, an electroactive inorganic/organic hybrid was obtained, namely, Ag-In-Zn-S/7-octyloxyphenazine-2-thiol, in which its organic part fully retained its electrochemical activity. © 2019 American Chemical Society.
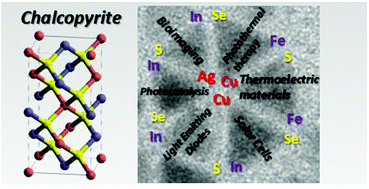
Moodelly D., Kowalik P., Bujak P., Pron A., Reiss P. Synthesis, photophysical properties and surface chemistry of chalcopyrite-type semiconductor nanocrystals
Journal of Materials Chemistry C 2019, 7, 38, 11665-11709
DOI: 10.1039/c9tc03875b
Abstract: Ternary and quaternary chalcopyrite-type nanocrystals of MIMIIIE2 and MIMIIIZnE2 type (MI = Ag, Cu; MIII = In, Fe; E = S, Se) and their core/shell structures have attracted a huge interest in the past decade because they combine size- and composition-tunable optical and electronic properties without relying on toxic elements such as Cd and Pb. These materials had been primarily studied as light harvesters in solar cell applications. However, in the form of colloidal nanocrystals, they exhibit remarkable photoluminescence properties in the visible and near infrared range. At the same time, the underlying emission mechanisms are distinctly different from those occurring in binary semiconductor quantum dots. Several models have been developed to explain the origin of the observed phenomena, in particular the line width broadening, large Stokes shifts and long photoluminescence lifetimes. This review first focusses on the current understanding of the optical properties of the title compounds. Next, an overview of the main synthesis methods is given, both in organic solvents and in the aqueous phase. In the second part, the surface chemistry and ligand exchange procedures are discussed. Finally, organic/inorganic hybrids of chalcopyrite nanocrystals with electroactive molecules are presented as well as their use in photovoltaics. © 2019 The Royal Society of Chemistry.
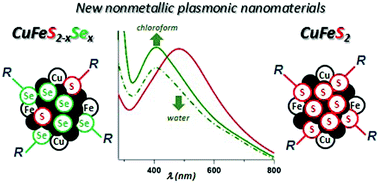
Kowalik P., Bujak P., Penkala M., Kotwica K., Kmita A., Gajewska M., Ostrowski A., Pron A. Synthesis of CuFeS2-xSex-alloyed nanocrystals with localized surface plasmon resonance in the visible spectral range
Journal of Materials Chemistry C 2019, 7, 21, 6246-6250
DOI: 10.1039/c9tc01242g
Abstract: Alloying of CuFeS2 and CuFeSe2 nanocrystals leads to a new plasmonic nanomaterial in which the plasmon resonance is hypsochromically shifted by ca. 80 nm as compared to the case of CuFeS2, i.e. from 486 nm to 408 nm. Preparation of CuFeS2-xSex facilitates, in addition, the exchange of the hydrophobic ligands for hydrophilic ones, rendering the nanocrystals dispersible in nonpolar and polar (aqueous) media. © 2019 The Royal Society of Chemistry.